Blood cultures are used to detect bacteraemia in children and infants. The validity of results obtained is entirely dependent on specimen collection. Inaccuracies can lead to diagnostic and therapeutic difficulties. Contamination, or false positive blood cultures, can lead to inappropriate treatment, increased hospital stays and hospital, laboratory and pharmacy costs [1] [2]. Alternatively a false negative blood cultures can incorrectly exclude bacteraemia, leading to inadequate microbial coverage [3].
The following factors are important in obtaining blood cultures:
Site Collection
Although easier to achieve, Everts et al found that cultures from intravascular catheters were more likely to be contaminated than venepuncture cultures [4]. Therefore cultures should not be obtained from peripheral or arterial lines unless upon insertion. Initial culture sites should be peripheral whilst trying to avoid sites immediately above peripheral lines. Additional blood cultures should be taken from a central line if line sepsis is suspected or there are difficulties obtaining peripheral access [1]. In the case of suspected line sepsis, peripheral cultures should be taken first followed by central cultures.
Number & Timing of Cultures
It is important that blood cultures are taken before microbial therapy is commenced, though this should not delay commencing treatment. Collection of blood cultures in relation to fever spikes has not been found to increase yield [1]. The maximum yield for blood cultures is 30 minutes to 2 hours before the onset of fever. The number of blood cultures has been shown to increase the detection of bacteraemia [5]. However, too many unnecessary cultures can lead to false positives.
Site Preparation
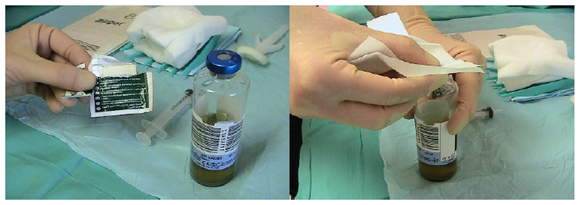
Skin should be thoroughly disinfected before cultures are taken. ChloraPrep (combination of 2% chlorahexidine gluconate and 70% isopropyl alcohol) has been shown to be effective in cutaneous antisepsis and is supported by the epic3 guidelines for preventing healthcare associated infections in NHS hospitals [6].
Optimum Blood Volume
There is a relationship between the volume of blood and detection of organisms with the volume of blood being the most important variable in improving yield of organisms from blood cultures [3]. The optimum blood volume needed for blood cultures to maximize yield of organisms is uncertain. Although small volumes may be adequate in neonates, Connell et al suggest a minimum of 0.5-1ml for infants [3]. It is thought that the level of bacteraemia is higher in infants, therefore less volume is necessary to detect this, however some studies have shown that there may be low levels of bacteraemia in infants and small children that may not be detected by smaller blood volumes [3]. Obviously in small children, the amount of blood sampled for cultures needs to be considered in context to the size of the child, but larger volumes do have greater yields.
Aerobic vs Anaerobic Cultures
Paediatric anaerobic infections are unusual and most anaerobes will grow in an aerobic culture bottle [1]. If obtaining adequate volume is difficult, then blood should be put into the aerobic bottle, rather than splitting the volume between the two bottles. Certain clinical situations where investigation for anaerobic bacteraemia is important include febrile neutropenia, necrotizing infections of head, neck, skin and soft tissues, necrotizing pneumonia, brain abscess, gynaecological infections and infections following colorectal surgery. Children or infants in septic shock with no obvious focus should also have anaerobic blood cultures [1].
Other Considerations
Agents that require discussion with microbiologists as they require special preparation include Cryptococcus neoformans, legionella, mycoplasma, helicobacter, leptospira and fungal pathogens.